Institute of Catalysis Research and Technology (IKFT)
The Institute of Catalysis Research and Technology was founded 2011. Its mission is to bridge the gap between fundamental and applied research and the development of new technologies and products in the field of catalysis and process technology of catalyzed processes. The focus of our work is the sustainable utilization of alternative feedstocks and their conversion into energy carriers intermediates. This includes the development of new catalytic systems based on a fundamental understanding of processes on a molecular level. The institute receives basic funding from the Helmholtz Association's program-oriented funding, largely in the Energy research field in the program Materials and Technologies for the Energy Transition.
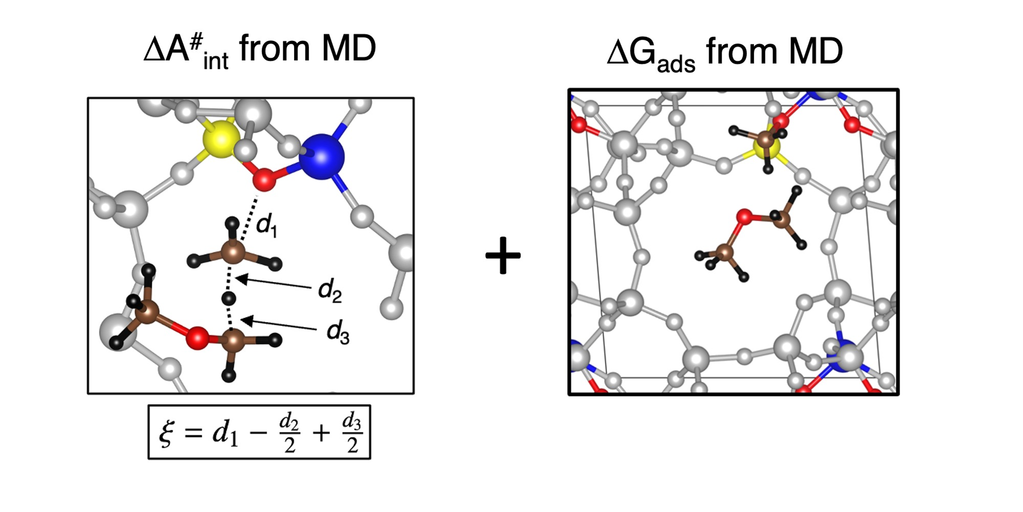
Es wurden genaue Molekulardynamik-Simulationen (MD) durchgeführt, um die Bildung von Formaldehyd in H-SSZ-13 zu untersuchen. Unter Verwendung eines kürzlich entwickelten Ansatzes wurden freie Aktivierungsenergien berechnet, in Verbindung mit genauen elektronischen Energien unter Einsatz von RPA-Berechnungen. Dies ermöglicht es, den Einfluss von Formaldehyd auf die Initiierung des MTO-Prozesses zu beurteilen.
mehr
We congratulate Wen Yanjun on successfully passing his doctoral examination on 17 March at the KIT Faculty of Chemical Engineering and Process Engineering.
He completed his doctoral research in Klaus Raffelt’s research group on the topic ‘Optimisation and exploration of catalysts for the hydrodeoxygenation upgrading of bio-oil through experiments and DFT calculations’.
more_rdax_98s.jpg)
The 23rd International Conference on “Fuels of the Future 2026” took place in Berlin in January, attended by over 620 participants from the worlds of politics, business, academia and industry associations.
more
On February 25, Constantin Fuchs successfully defended his dissertation entitled “Synthesis of aromatic-free gasoline and kerosene by means of heterogeneously catalyzed (co-)oligomerization of methanol-based olefins.” The supervisors were Professors Jörg Sauer and Reinhard Rauch.
Congratulations!
more_rdax_98s.jpg)
On 21 November, the Night of Science took place at KIT, and IKFT contributed to the vibrant program.
more
The DGMK event Hydrogen and Syngas - Platform for a sustainable future was jointly organized by the DGMK Divisions "Petrochemistry" and "Conversion of Carbon Carriers", the Division of Industrial Chemistry of the Società Chimica Italiana (SCI) and the ÖGEW Österreichische Gesellschaft für Energiewissenschaften.
moreVeranstaltungskalender
Understanding Activation of Small Molecules with Light: Photothermal Vs. Charge Carrier Effects in Photocatalysis
Understanding Activation of Small Molecules with Light:
Photothermal Vs. Charge Carrier Effects in Photocatalysis
Jennifer Strunk
Technical University of Munich
When photocatalysts are irradiated with high intensity light sources, it is expected that
the samples will also heat up, at least locally. Then, in addition to direct light-driven
reactions with excited charge carriers, more conventional thermal reactions are also
possible, similar to classical catalysis. This is especially the case when nanoparticles
of the coinage metals Cu, Ag and Au are involved, which show intense plasmon
absorption bands.
Here, two photocatalyst systems are presented in which we were able to clearly identify
photothermal effects as main reason for improved (photo)catalytic performance. For
the case of Au/TiO2, Anti-Stokes Raman spectroscopy was used to track temperature
changes in the anatase phase of TiO2, when the plasmon excitation of gold was
triggered with a green laser. Under these conditions, the titania reached temperatures
significantly exceeding 1000 K, which over time caused severe rutilization of titania.
Even under irradiation with common UV-Vis research light sources, rutilization of titania
was identified in Au/TiO2, so local temperatures above the rutilization threshold
(~850 K) can be reached in this case, too. At these temperatures, many catalytic
reactions of relevance reach thermodynamic equilibrium, so it is expected that thermal
effects are dominant for Au/TiO2, with little or no influence of photogenerated charge
carriers in either Au or TiO2.
In the second case, Cu/ZnO-based catalysts were considered for light-assisted
thermocatalytic CO2 valorization near ambient pressure. Both methanol synthesis and
CO formation by the reverse water gas shift were detected to be influenced by light
irradiation. The endothermic reverse water gas shift was always favored over
exothermic methanol synthesis, regardless of whether UV or visible light or both was
used. The behavior of forward and reverse reaction rate, together with detailed in situ
characterization of the material and the reaction mechanism, suggested that the light
simply caused the active sites, predominantly the copper, to be locally at much higher
temperature. The larger influence of visible light suggests a dominant influence of the
Cu plasmon absorption rather than band-gap excitation in ZnO.
Prof. Dr. Jennifer Strunk
Technical University of Munich (TUM)
