Institute of Catalysis Research and Technology (IKFT)
The Institute of Catalysis Research and Technology was founded 2011. Its mission is to bridge the gap between fundamental and applied research and the development of new technologies and products in the field of catalysis and process technology of catalyzed processes. The focus of our work is the sustainable utilization of alternative feedstocks and their conversion into energy carriers intermediates. This includes the development of new catalytic systems based on a fundamental understanding of processes on a molecular level. The institute receives basic funding from the Helmholtz Association's program-oriented funding, largely in the Energy research field in the program Materials and Technologies for the Energy Transition.

Die an der Fakultät für Chemieingenieurwesen und Verfahrenstechnik promovierenden Doktorand:Innen des IKFT haben in Annweiler am Trifels ein intensives Training zu Storytelling und Präsentationstechniken absolviert.
Link_more
Commissioned by BMW, KIT has prepared a widely acclaimed study on the potential of CO2-neutral fuels (CNF) based on biomass. The study reveals that renewable fuels represent a viable, scalable pillar for road transport without fossil CO in the EU.
more_rdax_1024x643_98.png)
The 5th edition of the Syngas Convention took place between the 22nd and the 25th of March in Cape Town, South Africa. The Care-O-Sene project, which is a cooperation between partners in South Africa and Germany for the development of sustainable aviation fuels, was strongly represented in the conference, with lectures from academia and industry.
more_rdax_1024x576_98.png)
Power‑to‑liquid (PtL) technologies are central to scaling sustainable aviation fuels (SAF) and reducing dependence on fossil‑based feedstocks. In our recent study, we benchmark two commercially relevant Fischer–Tropsch catalysts—20 wt.% Co on Al₂O₃ and TiO₂–SiO₂ supports—under industrial conditions. The TiO₂–SiO₂–supported catalyst delivers about 10% higher activity, higher C₅⁺ selectivity, and reduced methane formation.
more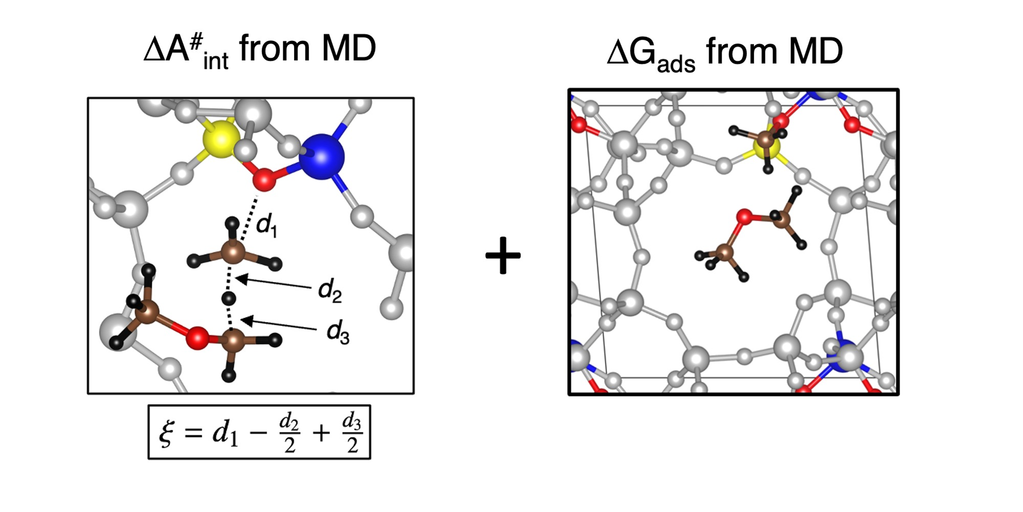
Accurate molecular dynamics (MD) simulations were performed to study the formation of formaldehyde in H-SSZ-13. Use a recently developed approach, apparent activation free energies were computed, in conjunction with accurate electronic energies using RPA calculations. This allows to judge the impact of formaldehyde in the initiation of the MTO process.
more
We congratulate Wen Yanjun on successfully passing his doctoral examination on 17 March at the KIT Faculty of Chemical Engineering and Process Engineering.
He completed his doctoral research in Klaus Raffelt’s research group on the topic ‘Optimisation and exploration of catalysts for the hydrodeoxygenation upgrading of bio-oil through experiments and DFT calculations’.
more